#2
Engelhardt B
Z Rheumatol 2000;59 Suppl 2:18-21
Max-Planck-Institut fur Physiologische und Klinische Forschung, W. G. Kerckhoff Institut, Abteilung Vaskulare Zellbiologie, Parkstr. 1, 61231 Bad Nauheim, Germany
PMID# 11155798; UI# 21028930
Abstract
Until recently, the Central Nervous System (CNS) has been considered to be an Immunologically privileged site, where the highly specialized Endothelial Blood-Brain Barrier (BBB) allows no entry of circulating Lymphocytes.
However, during inflammation in the CNS, MonoNuclear Cells readily gain access to the CNS Parenchyma.
The molecular mechanisms involved in Lymphocyte recruitment across the BBB have been mostly studied in Experimental AutoImmune EncephaloMyelitis (EAE), the prototype model for human Inflammatory DeMyelinating Diseases of the CNS such as Multiple Sclerosis (MS).
As Endothelial Cells actively participate in the regulation of Lymphocyte entry into various tissues, it is likely that the specialization of the BBB Endothelium extends to CNS-specific traffic signals involved in Lymphocyte recruitment.
Identification of the traffic signals mediating entry of Lymphocytes into the CNS is likely to be of great clinical importance, as blocking the responsible molecules possibly offers new specific routes of treatment of CNS Inflammatory Diseases.
Such a therapeutic regime, however, is not yet available. Rather, acute MS is often treated with high dose GlucoCorticoids.
Current evidence for a possible role of GlucoCorticoids in regulating the expression of Adhesion Molecules and thus inhibiting T-Cell recruitment across the BBB is discussed.
#3
Filippi M, Cercignani M, Inglese M, Horsfield MA, Comi G
Neurology 2001 Feb 13;56(3):304-311
Scientific Institute, Ospedale San Raffaele, Univ of Milan, NeuroImaging Research Unit, and Clinical Trials Unit, Dept of NeuroScience, Milan, Italy
PMID# 11171893
Abstract
Objectives
To quantify, using Diffusion Tensor Imaging (DTI), the tissue damage in lesions and Normal-Appearing White Matter (NAWM) from a large cohort of patients with MS.
And, to investigate the magnitude of the correlation between DTI-derived metrics and clinical disability.
Methods
Dual-Echo and DTI scans were obtained from 78 patients with Relapsing/Remitting, Secondary/Progressive, or Primary/Progressive MS and from 20 normal control participants. Post-contrast T1-weighted images were also obtained from the patients.
After creating Mean Diffusivity (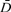 ) and Fractional Anisotropy (FA) images and image coregistration. ) and Fractional Anisotropy (FA) images and image coregistration.
FA values were measured for 4,846 Lesions:
Results
The average lesion was higher and the average lesion FA was lower than the corresponding quantities of the NAWM (p < 0.001).
The values of Enhancing and NonEnhancing Lesions were not different, whereas Enhancing lesions had lower FA (p < 0.001).
T1-HypoIntense lesions had higher and lower FA than T1-IsoIntense lesions (p < 0.001). NAWM of patients had higher and lower FA than White Matter of controls (p = 0.01).
Significant correlations were found between T1 and T2 lesion volume and and FA of lesions and NAWM.
In the overall patient sample, a moderate correlation was also found between lesion and the Expanded Disability Status Scale score (r = 0.28, p = 0.01).
However, the r value of this correlation was 0.48 in patients with Secondary/Progressive MS, whose Disability was also correlated with average lesion FA (r = -0.50).
Conclusions
The results of this study show that DTI is able to identify MS lesions with severe tissue damage and to detect changes in the NAWM.
They also indicate that DTI-derived measures are correlated with clinical disability, especially in patients with Secondary/Progressive MS, thus suggesting a role for DTI in monitoring advanced phases of the disease.
#4
Mirsattari SM, Johnston JB, McKenna R, Del Bigio MR, Orr P, Ross RT, Power C
Neurology 2001 Feb 13;56(3):317-323
Univ of Manitoba, Depts of Internal Medicine, Pathology, Pharmacology and Therapeutics, Manitoba, Winnipeg
PMID# 11171895
Abstract
Background
MS is common in people of Northern European ethnicity who live in Northern geographic areas; however, MS is rarely identified among Aboriginal peoples living in the same areas.
Objectives
To determine the prevalence, clinical features, HLA type, and Viral infections associated with MS among Aboriginals in Manitoba, Canada.
Methods
A retrospective study was performed in which the clinical features of all Aboriginal patients with MS together with HLA type and Human HerpesVirus-6, HIV-1, human T-Cell LymphoTropic Virus-1.
And, endogenous RetroVirus associated with MS (MSRV) infections were analyzed and compared with results from NonAboriginal patients with MS.
Results
Seven Aboriginals with MS were identified with a period prevalence among Aboriginals of 40:100,000.
Clinical features included Relapsing/Remitting (n = 6) or Primary/Progressive (n = 1) phenotypes with aggressive disease courses and frequent involvement of Optic Nerves and Spinal Cord (n = 5) compared with NonAboriginal patients.
Autopsy of one patient showed Necrosis and Eosinophil infiltrates in a Cervical Spinal Cord lesion and a DeMyelinated Optic Nerve.
Analysis of HLA alleles at the DRB1 and DQB1 loci indicated that the HLA types detected were common in Aboriginals, but there were no HLA alleles previously associated with the development of MS.
Analysis of the copy number of MRSV did not show differences among Aboriginals and NonAboriginals with or without MS.
Conclusions
Aboriginals of Algonkian background are at increased risk for an aggressive type of MS, resembling NeuroMyelitis Optica.
Which, is resistant to conventional MS treatments and occurs independently of HLA alleles previously associated with MS.
#5
Anti-DNA AntiBodies Are A Major Component Of The Intrathecal B-Cell Response In Multiple Sclerosis
Williamson RA, Burgoon MP, Owens GP, Ghausi O, Leclerc E, Firme L, Carlson S, Corboy J, Parren PW, Sanna PP, Gilden DH, Burton DR
Proc Natl Acad Sci USA 2001 Feb 13;98(4):1793-1798
The Scripps Research Institute, Depts of Immunology, Molecular Biology, and Neuropharmacology, La Jolla, CA 92037; and
Univ of Colorado Health Sciences Center, Depts of Neurology and Microbiology, Denver, CO 80262
PMID# 11172030
Abstract
Multiple Sclerosis (MS) is a Chronic Inflammatory DeMyelinating disease of unknown cause that afflicts the Central Nervous System.
MS is typified by a highly clonally restricted Antigen-driven AntiBody response that is confined largely to the Central Nervous System.
The major Antigenic targets of this response and the role of AntiBody in disease PathoGenesis remain unclear.
To help resolve these issues, we cloned the Ig G repertoire directly from active plaque and PeriPlaque regions in MS Brain and from B-Cells recovered from the CerebroSpinal Fluid of a patient with MS with subacute disease.
We found that high-affinity Anti-DNA AntiBodies are a major component of the Intrathecal IgG response in the patients with MS that we studied.
Furthermore, we show DNA-specific MonoClonal AntiBodies rescued from two subjects with MS as well as a DNA-specific AntiBody.
Rescued from an individual suffering from Systemic Lupus Erythematosus bound efficiently to the surface of Neuronal Cells and Oligodendrocytes.
For two of these AntiBodies, cell-surface recognition was DNA dependent.
Our findings indicate that Anti-DNA AntiBodies may promote important NeuroPathologic mechanisms in Chronic Inflammatory Disorders, such as MS and Systemic Lupus Erythematosus. |